درجة API
- تسمى أيضاً API gravity
- هى اختصار American Petroleum Institute معهد النفط الأمريكي.
- هي مقياس لحساب كثافة المواد البتروليه حسب معهد النفط الأمريكي.
- تعطى بالعلاقة:
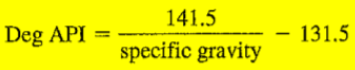
- حيث Specific gravity هي الوزن النوعى (الكثافة النسبية) وهى تساوى كثافة المادة مقسومة على كثافة الماء
أهمية مقياس API
(1) تصنيف النفط
- وفقاً لهذا المقياس يصنف النفط إلى درجات تتفاوت بينها وفق الكثافة النوعية بالنسبة إلى الماء إلى نفط ثقيل ومتوسط وخفيف.
- كلما كانت درجة API كبيرة كلما أصبح النفط خفيفاً والعكس صحيح
- تقاس الكثافة وفق مقياس الكثافة النوعية حسب معهد النفط الأمريكي API و يصنّف النفط إلى:
(أ) خفيف إذا كانت قيمتها أكبر من 31.10
(ب) متوسّط عند قيم بين 22.30 إلى 31.10
(جـ) ثقيل عندما قيمة API أقل من 22.30.
(2) حساب عدد البراميل من النفط الخام لكل طن متري
وذلك حسب العلاقة التالية:
